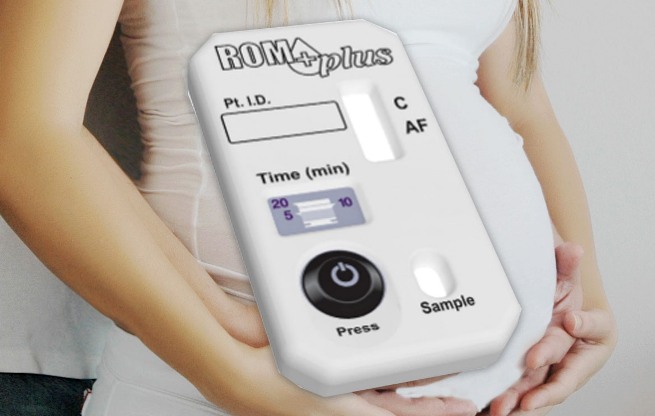
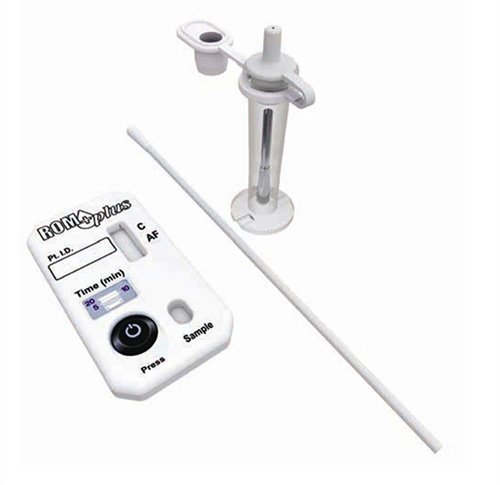
Rom Plus
Rupture of Membranes Test
ROM Plus® is unique in that it detects both Alpha-fetoprotein (AFP) and Insulin-like growth factor-binding protein 1 (IGFBP-1) using a monoclonal/polyclonal antibody approach–improving its sensitivity.
Timely and accurate diagnosis of PROM allows for gestational age-specific interventions designed to minimize the potential risk of complications to both mother and baby.

Dual Protein Markers: IGFBP-1 & AFP
- IGFBP-1 is detected early in first trimester and “plateaus” throughout the duration of the pregnancy
- AFP is detected at the end of the 2nd trimester/beginning of the 3rd trimester (< 37 weeks) where full confidence is required in the accurate diagnosis
Enquiry Form
Polyclonal & Monoclonal Antibody Approach:
- Reduces the chance of false negatives
- Provides a better “screening” method to detect the presence protein markers
- Improves the opportunity for denatured protein detection

What is sensitivity?
Sensitivity is the percentage probability that a positive test will occur in a patient who is really experiencing rupture of membranes, or the proportion of actual positives which are correctly identified as such. In a multi-site hospital study involving 285 patients, ROM Plus resulted in an overall sensitivity of 99.5%
What is ROM Plus' CLIA Classification?
ROM Plus test is not a waived test. It has been classified as Moderately Complex by CLIA (Clinical Laboratory Improvement Amendments).
Does blood contamination affect the test result?
ROM Plus test kits have been tested up to 10% blood concentration. In other words, it will function properly with trace amounts of blood in the sample, however, significant amounts of bloody discharge may cause the test to malfunction. ROM Plus is not recommended for use in these situations and it should be determined what is causing the bleeding.
How do I interpret ROM Plus test results?
If only a control line (C) is visible, the test result is negative (no IGFBP-1 and/or AFP were detected). If both the control line (C) and test line (AF) are visible, the test result is positive (IGFBP-1 and/or AFP was detected). If no lines are visible, or just the test line (AF) is visible, the test result is invalid and should be repeated. Darkness of the stripes may vary. The test is valid even if the stripes are faint. Do not interpret test results based on darkness of the stripes.
What are the ROM Plus storage and usage requirements for the kit, cassette and QC?
ROM Plus should be stored in a dry place at 4° to 37°C (40° to 99°F). DO NOT FREEZE. When stored in the foil pouch at the recommended temperature, the test is stable until the expiration date. Use ROM Plus within six (6) hours after opening foil pouch. Use ROM Plus within six (6) hours of collecting the vaginal swab sample and placing it into the buffer vial.
The lyophilized positive and negative controls can be stored in a dry place at room temperature until the expiration date. No refrigeration is required.
Do you have to use the timer in the cassette to run the test?
No. The timer that is built into the cassette is included for convenience and does not need to be used while running the test.
Will semen, urine or disinfectants interfere with test?
KY Gel, Surgilube, Tylenol, aspirin, Lever Soap, Noxzema cream, Pert Shampoo, human semen, urine and blood were tested as possible interference’s and were shown to be negative.
How often should external QC’s be run?
It is the company’s recommendation that external QC be completed for each new lot number or shipment of test material or if there is suspicion of improper storage and as required by regulatory or accrediting agencies.
What are the gestational age limits for ROM Plus?
ROM Plus does not have any gestational age limits.
Videos
